For US Healthcare Professionals
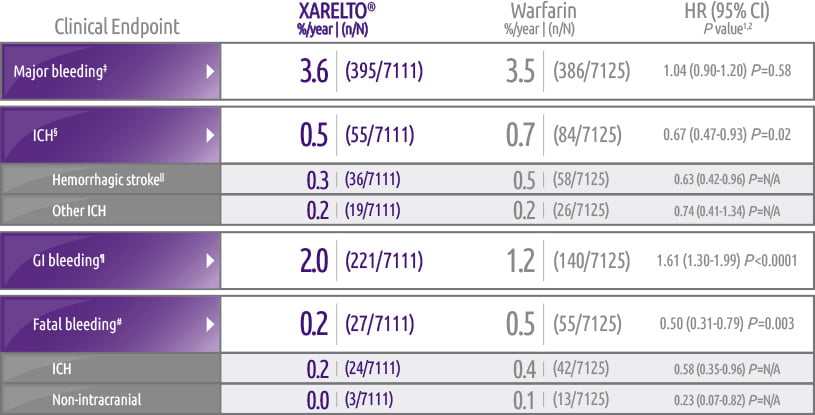
Bleeding events (on treatment plus 2 days) versus warfarin in ROCKET AF*
- Similar major bleeding†
- ICH and fatal bleeding rates
- GI bleeding with no difference in fatal GI bleeding